It is next to impossible to identify and divide the toxic substances in the food. But, sophisticated equipments like Gas Chromatographies are making the task easier for everyone and saving people from falling victim to toxicized food. There are numerous chromatography techniques for the same work. Yet, Gas chromatography is considered one of the best and most effective techniques.
While other chromatography techniques are also as widely used, Gas chromatography technique is most commonly used for fire residues, petroleum products, and other toxic substances.
Gas chromatography, in short, has evolved from identifying drugs in the blood to now different gases present in fire or alcohol etc. However, its application is not limited to these substances alone. Besides identifying different substances, Gas chromatography also divides them from the solutants.
There are very few teams who have taken interest in chromatography. Among them, there are even fewer teams who put in genuine efforts to provide genuine and authentic information to the audience. Argentoscientific is one such quality source for all information regarding analytical systems and chromatography.
Even though the team was formed in 2017, the efforts and attempts to start something that adds immense value goes back in time beyond 2017. If you want to learn more about analytical systems and chromatography, click for more information here.
Page Contents
Basics to know about Gas Chromatography columns
1. Gas Chromatography

Source: corning.com
Gas chromatography is nothing but an analytical method used to identify and divide selective or all chemical components from a given mixture. It can be used to identify certain substances, analyze the purity of particular components, and separate selected components from a particular mixture without disturbing the mixture. Although the name includes Gas, Gas chromatography can be used for all kinds of substances including gas, liquid, and solid.
2. Column of Gas chromatography
Columns of Gas chromatography can be defined as the heart and soul of the entire equipment. Although they look like test tubes in laboratories, they are simple column-like constructions made with thick and short glasses. Even stainless steel can also be used to make these columns. The reactions and interactions between the solutes and that the identification and separation of chemical components occur. Hence, columns are central and essential to the entire process of gas chromatography.
3. Limitations of columns
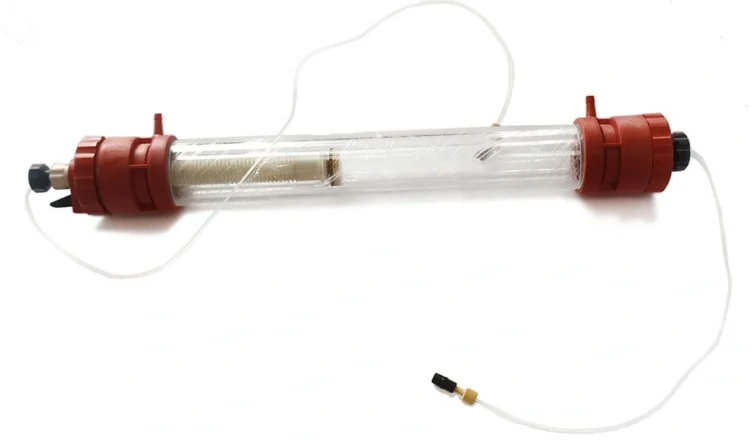
Source: galaklc.com
Column chromatography is considered one of the best and effective techniques of all methods. However, column chromatography still has its own fair share of limitations. It surely separates substances with decent accuracy. But, the entire process is a bit time-consuming. Also, the equipment of column-chromatography is considered expensive as compared to other analytical system equipment.
The power required for the entire separation process is low. It, hence, becomes complex to accurately apply such low levels of power. The entire process is automated. Although automated processes are more sort after, they cannot be regulated as required by the one who is performing the process. All these additional processes make the entire process too costly to bear especially for small amounts of samples, it is difficult to set up and a bit overwhelming.
4. Dry column
There are certain unknown factors that make it difficult for beginners to understand the functionality and errors involved in the process. For example, columns should be moist before and during the process according to rules and prerequisites. Hence, it is essential to make sure that the columns are not dry. It is better to check the columns before including them in the equipment.
Dry columns may make cracks in the silica component. Dried and cracked silica components cause poor identification and separation of components. If you allow the solvent level to run below the silica level, it is an indication that your column goes dry and therefore, impacts the separation process.
5. Type of columns

Source: galak-tech.en.made-in-china.com
There are different types of columns. Not all columns allow or facilitate different processes. For example, for analytical separation processes, capillary columns are considered the best choice.
They provide sharp peaks and an effective separation process. While some chemical components are resistant to external factors, a few others are less resistant and sensitive to external changes. In such cases, capillary columns are best to provide high-sensitive procedures.
6. Efficiency
There are numerous gas chromatography processes available for similar purposes. However, column chromatography is the best and most efficient of all. This conclusion has been deducted after much deliberation, empirical analysis, and laboratory assessments.
Because column-chromatography provides the purest of chemical separations and the values too. The errors and variables involved in this process are almost next to negligible. Hence, the efficiency, reliability, and credibility of column-chromatography are high as compared to the other processes.
7. Exit

Source: wikipedia.org
Identification and separation of chemical components is just one part of the entire process. There are numerous other parts of the procedure involved in this entire process. As substances flow in the column, molecules adsorb to the column. But, the process is reversibly and automatic. Some substances interact weakly, whereas others interact strongly.
The weakly interacted substances exit the column really fast as compared to the ones that interacted strongly. This process of exiting the column somewhat influences the complexity and time taken to finish the entire process.
8. Buffer
In contrast to other processes, the buffer is extremely essential in the case of columns. The addition of a buffer maintains and regulates the level of pH in the columns, which is necessary. The ionizable components are sensitive to mobile phase pH. Hence, adding buffer controls and neutralizing it all.
Conclusion
It is difficult to imagine gas chromatography without the columns. Because columns play a crucial role in the entire process of gas chromatography. The columns in gas chromatography equipment are also called stationary phases. Even though the entire process and mobility of substances occur in columns, the columns remain unmoved and stationary. Hence, they are named stationary phases in the entire equipment or process.
These are just a few basic things to know about columns in gas chromatography. There are a lot more things to learn and know. However, these are the most basic things that are essential to start working with and handling the chromatography equipment.





